
HTRF Human Total SHP1 Detection Kit, 500 Assay Points


HTRF Human Total SHP1 Detection Kit, 500 Assay Points






The Total SHP1 kit is designed to monitor the expression level of SHP1, and can be used as a normalization assay for the Phospho-SHP1 (Tyr564) kit.
| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |
The Total SHP1 kit is designed to monitor the expression level of SHP1, and can be used as a normalization assay for the Phospho-SHP1 (Tyr564) kit.



HTRF Human Total SHP1 Detection Kit, 500 Assay Points



HTRF Human Total SHP1 Detection Kit, 500 Assay Points



Product information
Overview
The Total SHP1 cellular assay monitors the expression level of SHP1, and can be used as a normalization assay with our phospho-SHP1 Y564 kit. Because these phospho and total SHP1 assays are compatible, the two kits can be used in parallel on the same lysates.
Many cancer cells overexpress checkpoint inhibitor ligands such as PD-L1. PD-L1 binds its counterpart checkpoint inhibitor receptor PD1, present at the surface of T lymphocytes. In turn, the PD1-PDL1 complex recruits and activates inhibitory effectors, such as SHP1 or SHP2. These two phosphatases, which are phosphorylated on Tyr564 and Tyr542 respectively by the kinase Lck, trigger the dephosphorylation of signaling proteins such as ZAP-70 or SLP-76, involved in the T cell activation pathway. Finally, activated SHP1 and SHP2 participate in T cell inactivation.
Preventing the activation of SHP1 and/or SHP2 by small molecule inhibitors is believed to contribute to restoring the immune response against tumors.
Specifications
| Application |
Cell Signaling
|
|---|---|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 2
Lysis Buffer 3
Lysis Buffer 4
Lysis Buffer 5
|
| Molecular Modification |
Total
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Infectious Diseases
Oncology & Inflammation
|
| Unit Size |
500 Assay Points
|
Video gallery

HTRF Human Total SHP1 Detection Kit, 500 Assay Points

HTRF Human Total SHP1 Detection Kit, 500 Assay Points

How it works
Total SHP1 assay principle
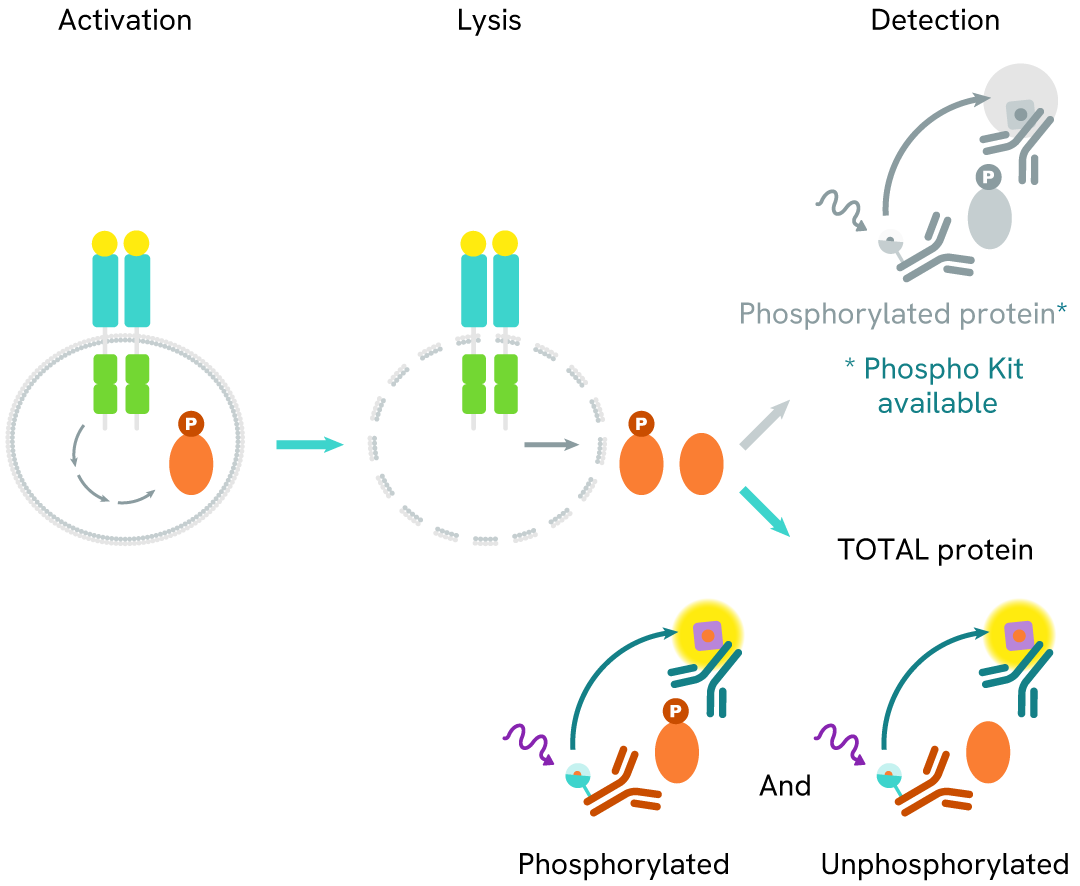
The Total SHP1 assay measures the expression level of SHP1 independently of its phosphorylation state. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer. The Total SHP1 assay uses 2 labeled antibodies, one with a donor fluorophore, the other with an acceptor. Both antibodies are highly specific for a distinct epitope on the protein. In presence of SHP1 in a cell extract, the addition of these conjugates brings the donor fluorophore into close proximity with the acceptor and thereby generates a FRET signal. Its intensity is directly proportional to the concentration of the protein present in the sample, and provides a means of assessing the protein's expression under a no-wash assay format.

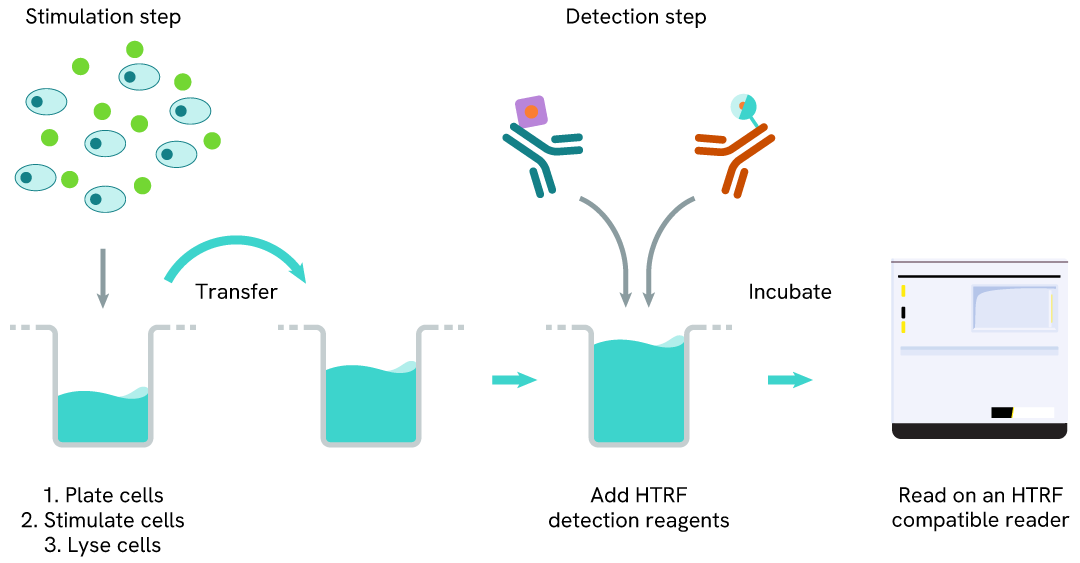
Total SHP1 two-plate assay protocol
The two-plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates to a 384-well low volume detection plate before the addition of Total SHP1 HTRF detection reagents. This protocol enables the cells' viability and confluence to be monitored.
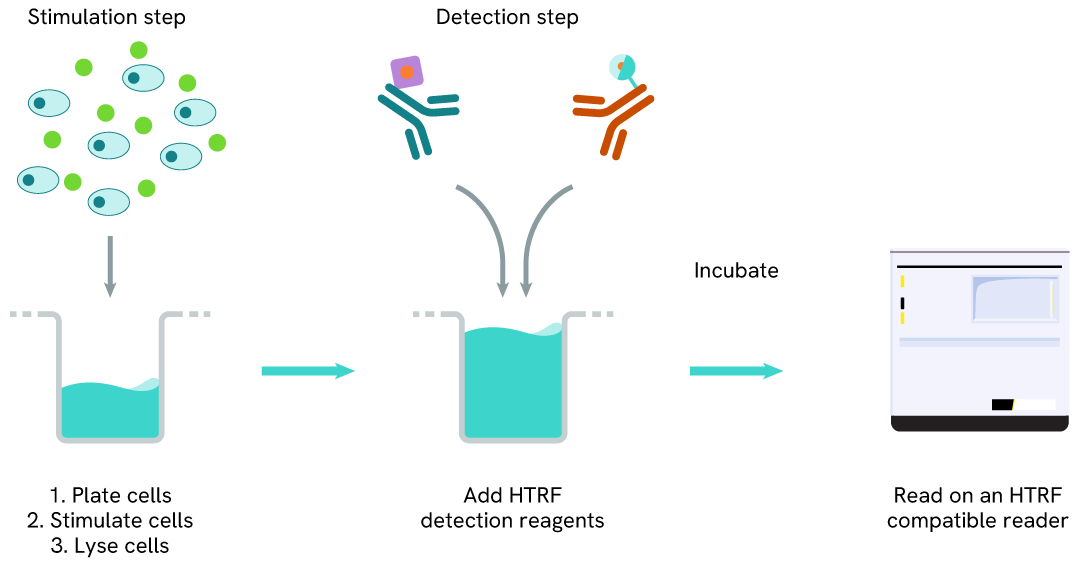
Total SHP1 one-plate assay protocol
Detection of Total SHP1 with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required. This HTS designed protocol enables miniaturization while maintaining robust HTRF quality.
Assay validation
Pharmacological validation using the Lck inhibitor, saracatinib, in Jurkat T-cells
Human Jurkat suspension cells were plated at 100,000 cells/well in a 96-well half area plate, and incubated for 24 h at 37°C, 5% CO2, with increasing concentrations of Saracatinib. Before lysis, Jurkat cells were incubated 30min with Pervanadate (30 µM), followed by the addition of 10µL of supplemented lysis buffer 4X. After 30min lysis at RT under gentle shaking, 16 µL of lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF phospho-SHP1 (Tyr564) or total SHP1 detection reagents were added. The HTRF signal was recorded after an overnight incubation.
As described elsewhere, a dose dependent inhibition of SHP1 Tyr564 phosphorylation was obtained following treatment with Saracatinib, whereas the SHP1 expression level remained stable under the same experimental conditions.

HTRF total-SHP1 cellular assays compared to Western Blot
The human Jurkat cell line was seeded in a T175 flask, and incubated at 37°C, 5% CO2. The cells were then treated with Pervanadate (30 µM) for 30 min before lysis.
Serial dilutions of the cell lysate were performed in the supplemented lysis buffer, and 16µL of each dilution were transferred into a low volume white microplate before the addition of 4µL of HTRF total-SHP1 detection reagents. Equal amounts of lysates were used for a side by side comparison between HTRF and Western Blot.
For HTRF total-SHP1 assay and for Western Blot, 5,000 cells/well were sufficient to detect a signal, demonstrating the similar sensitivity of the two methods.
Simplified pathway
Function and regulation of SHP1
SHP1 (also known as tyrosine-protein phosphatase non-receptor type 6, PTPN6) is a tyrosine phosphatase mainly expressed in hematopoietic cells, activated by Lck and recruited by cellular surface receptors. SHP2 (also known as tyrosine-protein phosphatase non-receptor type 11) is ubiquitously expressed in hematopoietic or non-hematopoietic cells. Although SHP2 negatively regulates T cell activation, SHP2 is positively involved in ERK activation in response to growth factors such as PDGF or FGF.
In T lymphocyte cells, SHP1 and 2 are recruited by immune checkpoint inhibitors, thereby participating in the suppression of the TCR signaling pathway. SHP1 and 2 interact with PD1 ITIM domains and are phosphorylated and activated by the Lck kinase. Activated SHP1 and 2 phosphatases lead to the dephosphorylation of key TCR signaling effectors, such as ZAP70 or SLP76, which are required for T-cell proliferation and function.
Resources
Are you looking for resources, click on the resource type to explore further.
Discover the versatility and precision of Homogeneous Time-Resolved Fluorescence (HTRF) technology. Our HTRF portfolio offers a...


How can we help you?
We are here to answer your questions.






























