HTRF Human Total CHK-2 Detection Kit, 500 Assay Points

HTRF Human Total CHK-2 Detection Kit, 500 Assay Points




| Feature | Specification |
|---|---|
| Application | Cell Signaling |
| Sample Volume | 16 µL |









Product information
Overview
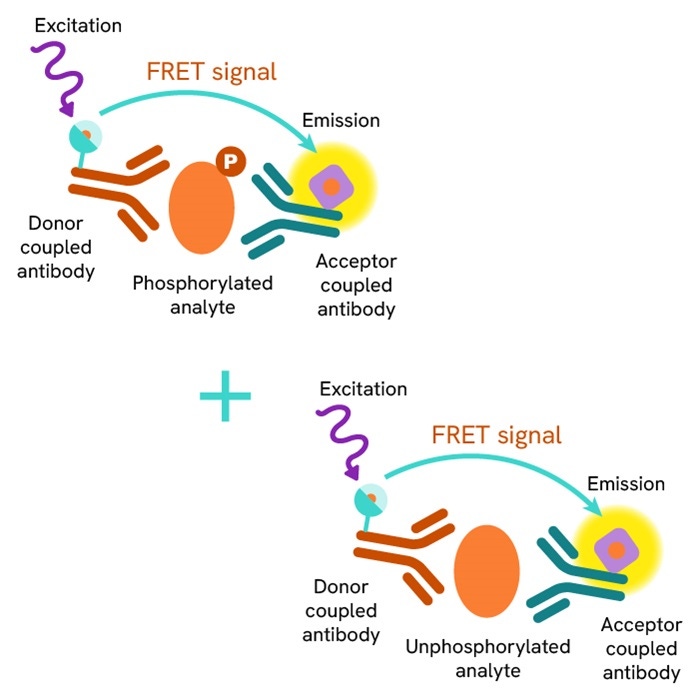
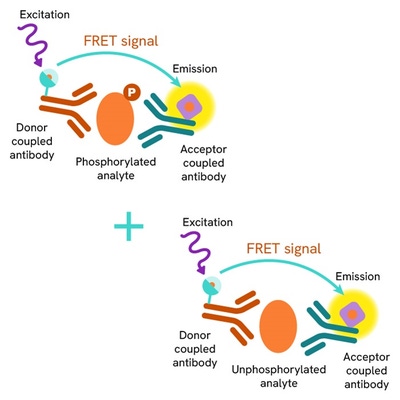
The Total-CHK2 cellular assay kit is designed to monitor the expression level of CHK2, whether phosphorylated or unphosphorylated.
It is compatible with our Phospho-CHK2 thr68 kit, and enables the analysis of phosphorylated and total proteins from a single sample for a better readout of ATM/CHK2 pathway.
How it works
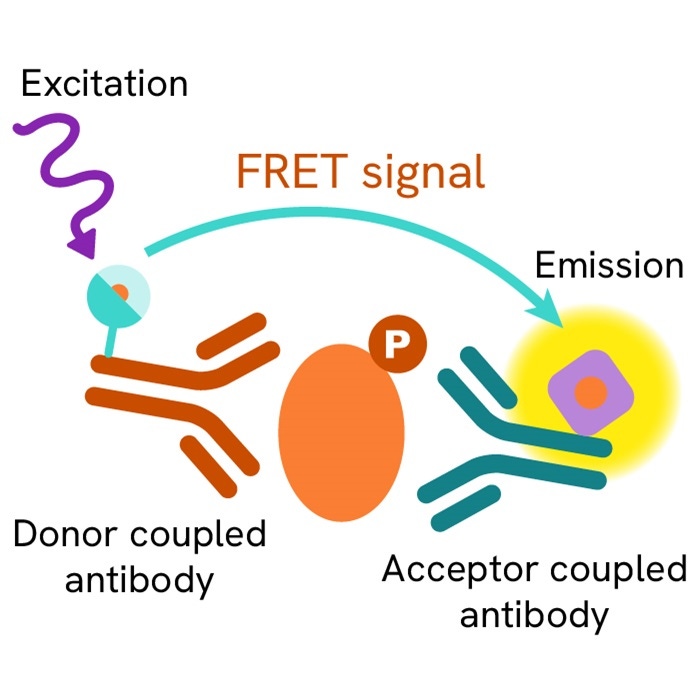
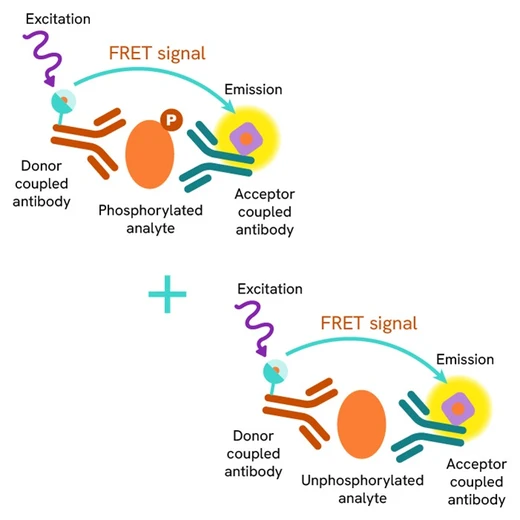
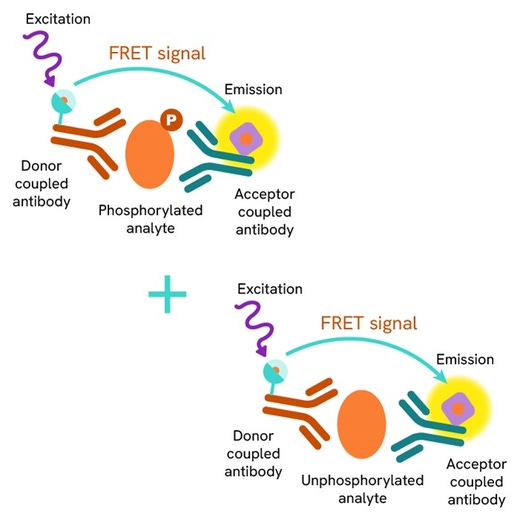
HTRF Total CHK2 assay principle
The Total-CHK2 assay quantifies the expression level of CHK2 in a cell lysate. Unlike Western Blot, the assay is entirely plate-based and does not require gels, electrophoresis, or transfer.The Total-CHK2 assay uses two labeled antibodies: one coupled to a donor fluorophore, the other to an acceptor. Both antibodies are highly specific for a distinct epitope on the protein. In presence of CHK2 in a cell extract, the addition of these conjugates brings the donor fluorophore into close proximity with the acceptor and thereby generates a FRET signal. Its intensity is directly proportional to the concentration of the protein present in the sample, and provides a means of assessing the protein’s expression under a no-wash assay format.

HTRF Total-CHK2 two-plate assay protocol
The 2 plate protocol involves culturing cells in a 96-well plate before lysis, then transferring lysates to a 384-well low volume detection plate before the addition of the Total-CHK2 HTRF detection reagents.
This protocol enables the cells' viability and confluence to be monitored.

HTRF Total-CHK2 one-plate assay protocol
Detection of total CHK2 with HTRF reagents can be performed in a single plate used for culturing, stimulation, and lysis. No washing steps are required.
This HTS-designed protocol enables miniaturization while maintaining robust HTRF quality.

Assay validation
Neocarzinostatin effect on total and phospho Thr 68 CHK2 assay
Human HEK293 cells were plated in 96-well culture-treated plate (100,000 cells/well) in complete culture medium, and incubated overnight at 37°C,5%CO2. Cells were treated with a dose-response of Neocarzinostatin 2h at 37 °C, 5% CO2. Cells were then lysed with 50 µl of supplemented lysis buffer #1 (1X) for 30 min at RT under gentle shaking. After cell lysis, 16 µL of lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Phospho CHK2 (Thr 68) or Total-CHK2 detection reagents were added. The HTRF signal was recorded after an overnight incubation at room temperature.
As expected, Neocarzinostatin induced single and double strand DNA damages leading to a dose-dependent increase of CHK2 phosphorylation, without effect on the expression level of the CHK2 total protein.

Effect of compounds inducing DNA damage on CHK2 phosphorylation and total protein
Human HEK293 cells were plated in 96-well culture-treated plate (100,000 cells/well) in complete culture medium, and incubated overnight at 37°C,5%CO2. Cells were treated with a dose-response of Neocarzinostatin, hydroxyurea, Doxorubicin and Etoposid, 2h at 37 °C, 5% CO2. The medium was then removed, and the cells were lysed with 50 µl of supplemented lysis buffer #1 (1X) for 30 min at RT under gentle shaking. After cell lysis, 16 µL of lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Phospho CHK2 (Thr 68) or Total-CHK2 detection reagents were added. The HTRF signal was recorded after an overnight incubation at room temperature.
The different compounds show different response. Neocarzinostatin, doxorubicin and etoposide known to induce double strand breaks (DSB) lead to phosphorylation of CHK2. On the other hand, Hydroxyurea that preferably induces single strand breaks (SSB) displays a partial CHK2 phosphorylation with weak potency. The EC50 of neocarzinostatin, doxorubicin, etoposide and hydroxyurea were respectively evaluated at 0.1 µM, 1.5 µM, 0.4 µM and 0.6 mM respectively.
Moreover, EC80 of Neocarzinostatin was evaluated at 0.3 µM and this concentration was used to assess inhibitors of ATM/CHK2 Pathway.
On the other hand, none of the 4 compounds affects the expression level of the CHK2 total protein.


Effect of ATR/CHK1 or ATM/CHK2 pathways inhibitors on HTRF Phospho Thr 68 and total CHK2 kits
Human HEK293 cells were plated in 96-well culture-treated plate (100,000 cells/well) in complete culture medium, and incubated overnight at 37°C,5%CO2. Cells were treated with a dose-response of 3 inhibitors of ATR or ATM pathway for 2h at 37 °C, 5% CO2. The cells were then treated with 0.3 µM of Neocarzinostatin (EC80) for another 2h at 37 °C, 5% CO2. The medium was removed, and the cells were then lysed with 50 µl of supplemented lysis buffer #1 (1X) for 30 min at RT under gentle shaking. After cell lysis, 16 µL of lysate were transferred into a 384-well low volume white microplate and 4 µL of the HTRF Phospho CHK2 (Thr 68) or Total-CHK2 detection reagents were added. The HTRF signal was recorded after an overnight incubation at room temperature.
Caffeine is known as a mild ATR/ATM pathway inhibitor; UCN-1 as a potent CHK1 inhibitor and KU55933 as a selective ATM pathway inhibitor. As expected, UCN-1 has no effect on CHK2 phosphorylation. Caffeine shows a decrease of CHK2 phosphorylation with a weak potency. KU55933 allows a full inhibition of CHK2 phosphorylation with a higher potency (IC50 : 1 µM).
These 3 tested compounds don’t affect the expression level of the CHK2 total protein.


HTRF Total CHK2 Assay compared to Western Blot
HEK293 cells were cultured in complete medium at 37°C, 5% CO2 in a T175 flask to confluency.
After medium removal, the cells were lysed with 3 mL of supplemented lysis buffer #1 (1x) for 30 min at RT under gentle shaking.
Serial dilutions of the cell lysate were performed using supplemented lysis buffer #1 (1x), and 16µL of pure sample and each dilution were transferred into a 384-well small volume microplate, before the addition of 4µL of HTRF Total CHK2 detection reagents. Signals were recorded overnight.
Equal amounts of lysates were loaded into a gel for a side by side comparison between HTRF and Western Blot.
In these conditions, the HTRF total CHK2 assay is as sensitive as the Western Blot.

Simplified pathway
ATM/CHK2 and ATR/CHK1 signaling pathways
Double-strand breaks (DSBs) or single-strand breaks (SSBs) are among the most deleterious lesions that threaten genome integrity. These damages can be induced by the effect of cellular metabolites or by DNA-damaging agents such as genotoxics compounds, chemotherapeutic agents, ultraviolet (UV) irradiation or ionizing radiation.
DNA damage response (DDR) is mainly controlled by ataxia telangiectasia mutated (ATM) and ataxia telangiectasia and Rad3-related (ATR), two members of the phosphoinositide 3-kinase (PI3K)-related kinase (PIKK) protein kinase family.
In reponse to DNA damage (DSBs), ATM–Chk2 pathway and replication checkpoint responses are activated that mediate G1/S checkpoints to arrest cell cycle progression and allow extra time for DNA repair.
In response to DNA DSBs, the activation of ATM results in the phosphorylation of the checkpoint kinase Chk2. Chk2 is a stable protein expressed throughout the cell cycle, it appears to be largely inactive in the absence of DNA damage. Its activation involves dimerization and autophosphorylation (Thr383 and Thr 387) and induces the activation of the downstream signal effectors such as the tumor suppressor protein p53, CdC25C and control cdk2/CyclinA activity.
Chemical inhibition of Chk2 during radiation might protect sensitive tissues such as lymphocytes or intestinal epithelium from the side effects of radiotherapy or drugs that cause DSBs. The critical issue here would be to identify suitable inhibitors of Chk2 and test whether this strategy could be applied without increasing the incidence of tumors.
Identification of new small molecule inhibitors of Chk2 and design and validation of novel strategies of checkpoint modulation, combined with the traditional radiation and chemotherapy modalities, hold promise for improved treatment of cancer.

Specifications
| Application |
Cell Signaling
|
|---|---|
| Automation Compatible |
Yes
|
| Brand |
HTRF
|
| Detection Modality |
HTRF
|
| Lysis Buffer Compatibility |
Lysis Buffer 1
Lysis Buffer 4
|
| Molecular Modification |
Total
|
| Product Group |
Kit
|
| Sample Volume |
16 µL
|
| Shipping Conditions |
Shipped in Dry Ice
|
| Target Class |
Phosphoproteins
|
| Target Species |
Human
|
| Technology |
TR-FRET
|
| Therapeutic Area |
Oncology & Inflammation
|
| Unit Size |
500 assay points
|
Video gallery
Resources
Are you looking for resources, click on the resource type to explore further.
This guide provides you an overview of HTRF applications in several therapeutic areas.
SDS, COAs, manuals and more
Are you looking for technical documents related to the product? We have categorized them in dedicated sections below. Explore now or request your COA/TDS, SDS, or IFU/manual.
- LanguageEnglishCountryUnited States
- LanguageFrenchCountryFrance
- LanguageGermanCountryGermany
- Resource TypeManualLanguageEnglishCountry-


Recently viewed

How can we help you?
We are here to answer your questions.